Thanks to the entire Medical team (Dr Bachir Athmani, Dr Marc Abécassis, Dr Fadel, Dr Miguel Cortes) and the IT team (Richard Coffre, Flavien Palero, Laetitia Bernard-Granger) as well as to Adrien Coffre for his excellent graphic designer work.
💢💢💢 Preventive strategy: the 8 main “3M3T-AV” measures 💢💢💢
In the current environment, a small positive note to enlighten your weekend, the celebration of our memo at the end of February 2020, on the 8 main preventive strategic measures concerning the Covid: “3M3T” (updated by adding AVé “).
This memo, which I published on CovidMinute a year ago, has the merit of being effective, consistent … and translatable into all Western languages (=> widely distributed and used).
We have added a well-known and up-to-date visual aid for educational purposes (more difficult with the Smurfs or Asterix, but we are already working on it!).
Individual measures: “3M”
Mask :
Note that China delivered to the world more than 24 billion masks in 2020, or 3 per inhabitant of the planet.
Main: “3M” are particularly effective, as shown by the drastic reduction in viral diseases (influenza, viral gastroenteritis, etc.) usual in 2020.
Metre :
This variable geometry measure depending on the country (1-2 meter) has had a terrible impact on the “proximity” sectors …
Collective measures (secondary prevention): “3T”
Test:
Note that the Indonesian test (“GetNose C19”) based on artificial intelligence, recognizes volatile organic C + components, and has been valid in this country (sensitivity at 95% equivalent to the PCR test). France is also in the development phase of a clinical trial for C + sniffer dogs …
Tracking:
Asia has used digital tracking a lot (telephone, bank card, etc.) which is certainly liberticidal … but much more effective than “Western” mechanical tracking
Triage (= see for memo => = isolation):
The entire preventive strategy is based on the speed of execution (see 3-day time limit for test results!), The outcome of which is temporary isolation (see family cases ++), which has proven to be much more effective in Asia.
Collective measures (primary prevention): “AVe”
Airport (access to the territory): note the rule of the “3 ANDs” in China versus the “3 ORs” in Europe.
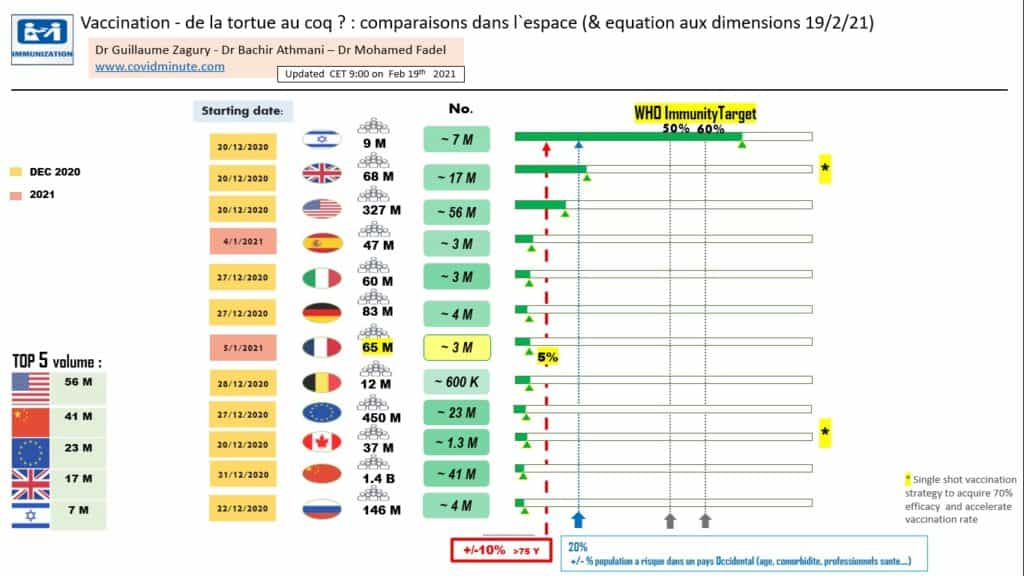
Vaccine: This is the current issue, given its implications. Note that it has been made compulsory in Indonesia, and that the digital vaccine passport will be unavoidable for access to territories or to certain cultural and sporting activities …
For our part, we consider that as soon as the 20% of those most at risk will be vaccinated (probably at the end of April in France), we will have to immediately relaunch our economy: we will have done the maximum for people at risk, and hoping that our health system will not be saturated.
Note that in terms of COVID GEOPATHOLOGY, through these 8 items, viral RNA has made it possible to update the DNA of our different Societies. This is what I have made every effort to show since 1/28/2020, when I started to accompany you in this epidemic, initially on RS Wechat in China, then on Covidminute.
💢💢💢Small complement 💢💢💢
Do not hesitate to let us know if you enjoy this type of presentation (“serious without taking yourself seriously”), we have reservations, and are sharing several more on the site www.covidminute.com
Next meeting: anniversary of COVIDSCORE created in early 2020 (= quantified assessment of the different countries on “3M3T” aspects), and follow-up Israel (small country but great champion of vaccination).
—————————————————————–
If you appreciate our “100% original” visuals, do not hesitate to offer your Doctor the “Best-Seller”: Médi Mémo Minute (more than 30,000 books sold)
The gift your Doctor will not forget …
—————————————————–As for a year, 3M (Mask-Hand-Meter) and these days for a “special Israel”, laboratory of the Planet …
Hoping that 2021 will bring us vaccines, and the joy of regaining our “freedom”,
Dr Guillaume Zagury
Specialist in International Public Health (DES-Paris)
HEC
Practitioner
Consultant in Medical Innovations (France, Israel, China)
In China for 20 years
@: Guillaumez888@hotmail.com
The alt attribute of this image is empty, its file name is folder.png.